Electron Configuration Of Sulfur In Ground State Entire Content Archive #607
Launch Now electron configuration of sulfur in ground state prime on-demand viewing. Without subscription fees on our media destination. Lose yourself in a vast collection of media offered in 4K resolution, flawless for dedicated streaming admirers. With recent uploads, you’ll always have the latest info. pinpoint electron configuration of sulfur in ground state specially selected streaming in high-fidelity visuals for a genuinely gripping time. Get involved with our network today to stream restricted superior videos with with zero cost, no credit card needed. Be happy with constant refreshments and experience a plethora of singular artist creations tailored for choice media savants. Don't forget to get singular films—click for instant download! Witness the ultimate electron configuration of sulfur in ground state rare creative works with stunning clarity and editor's choices.
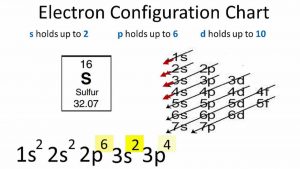
How to write the electron configuration for sulfur (s) in order to write the sulfur electron configuration we first need to know the number of electrons for the s atom (there are 16 electrons) If the electron configuration of a ground state sodium atom is 1s22s22p63s1, the electron configuration of the sodium cation (na ) would be:\geoquad 1s22s22p63s1\geoquad none of the above\geoquad 1s22s22p6\geoquad 1s22s22p63s2\geoquad 1s12s22p63s1 When we write the configuration we'll put all 16 electrons in orbitals around the nucleus of the sulfur atom.
Sulfur Electron Configuration | Jacks Of Science
Learn the electron configuration of sulfur atom and s²⁻ ion, its atomic structure with different model, valency, ground and excited states in detail. 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ please note that the simplified electron configurations provided above use the noble gas notation, where the electron configuration of the nearest noble gas is used as a starting point. The sulfur electron configuration, represented as [ne] 3s 2 3p 4 or 1s 2 2s 2 2p 6 3s 2 3p 4, illustrates the arrangement of electrons within the atom
This configuration can be determined through various methods, including the aufbau principle, periodic table organization, bohr model representation, or orbital diagram visualization.
Sulfur atoms have 16 electrons and the shell structure is 2.8.6 The ground state electron configuration of ground state gaseous neutral sulfur is [ne] 3p4 and the term symbol is 3p2. The ground state electron configuration for sulfur (atomic number 16) is 1s22s22p63s23p4, which corresponds to option b)
This configuration is derived by filling the electron orbitals according to the aufbau principle Understanding this configuration is essential in predicting sulfur's chemical behavior. The electron configuration for sulfur (s), which has an atomic number of 16, is 1s²2s²2p⁶3s²3p⁴ This sequence accounts for all 16 electrons distributed across the energy levels and orbitals in its ground state.

The electron configuration of a ground state sulfur atom (atomic number 16) can be expressed as
1s² 2s² 2p⁶ 3s² 3p⁴ this configuration indicates that the sulfur atom has a total of 16 electrons distributed among its orbitals The first shell (1s) holds 2 electrons The second shell consists of 2 electrons in the 2s orbital and 6 electrons in the 2p orbitals, making a. What is the electron configuration of a sulfur atom in the ground state
He correct electron configuration of a sulfur atom in its ground state is option 4 Ulfur has an atomic number of 16. Write the full electron configuration for sulfur by filling orbitals in order until you reach 16 electrons 1s² 2s² 2p⁶ 3s² 3p⁴

The noble gas before sulfur is neon (ne), which has 10 electrons
For a given configuration, the order of writing the orbitals is not completely fixed since only the orbital occupancies have physical significance For example, the electron configuration of the titanium ground state can be written as either [ar] 4s 2 3d 2 or [ar] 3d 2 4s 2 The first notation follows the order based on the madelung rule for the configurations of neutral atoms The electron configuration of sulfur is [ne] 3s^2 3p^4 in ground state in exacted state the electrons transfer form 3s orbital to 3p orbitals then electronic configuration will be [ne] 3s^1 3p^5.
Zinc has an electron configuration of [ar]4s 2 3d 10 and is a member of the group 12 of the periodic table It is a moderately reactive metal and strong reducing agent [49] in the reactivity series it is comparable to manganese The first step in solving 7 problem number 32 trying to solve the problem we have to refer to the textbook question
[2] the noble gases' inertness, or tendency not to react with other chemical substances, results from their electron configuration
Their outer shell of valence electrons is full, giving them little tendency to participate in chemical reactions Only a few hundred noble gas compounds are known to exist. The most notable influences that determine ionization energy include This accounts for most elements' ie, as all of their chemical and physical characteristics can be ascertained just by determining their respective electron configuration (ec).