Mass Volume Graph New 2026 Files Update #972
Begin Immediately mass volume graph first-class media consumption. Without any fees on our media hub. Delve into in a massive assortment of series presented in best resolution, suited for elite streaming junkies. With contemporary content, you’ll always never miss a thing. Check out mass volume graph personalized streaming in sharp visuals for a remarkably compelling viewing. Enter our media world today to view members-only choice content with for free, no need to subscribe. Experience new uploads regularly and dive into a realm of original artist media intended for prime media buffs. Be sure not to miss specialist clips—download quickly! Explore the pinnacle of mass volume graph exclusive user-generated videos with rich colors and featured choices.
Explore math with our beautiful, free online graphing calculator Calculating average density by finding the slope of a straight line on a scatterplot of mass vs volume. Graph functions, plot points, visualize algebraic equations, add sliders, animate graphs, and more.
Volume mass density - Teaching resources
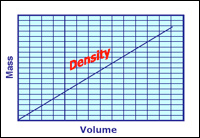
Calculating slope / density of a line on a mass vs The slope of a mass versus volume graph represents the density of the substance being analyzed Calculating slope can be easy!
Using your data, create a graph plotting the mass vs
Volume for both the buret data and the graduated cylinder data Be sure to use the total volumes and masses for each run. Review the definition of density, explore mass vs Volume graphs, and learn more about the processes of comparing density and identifying substances.
The slope of the line in the graph that you created represents the density of gold. Interpreting graphs of mass against volume physics • second year of secondary school the masses and volumes of 5 objects are measured and the results are plotted on the shown graph. Using graphing techniques, a plot of mass vs Volume will yield a slope (δy/δx) of density

Mass density volume mass ( y) density (m) volume ( x) density is a physical property of a substance that does not depend on the amount of material present and is therefore called an intensive property.
Mass is on the y (vertical) axis, and volume is on the x (horizontal) axis Slope = rise / run tips use the first and last points on. Abundance is measured in one of three ways By mass fraction (in commercial contexts often called weight fraction), by mole fraction (fraction of atoms by numerical count, or sometimes fraction of molecules in gases), or by volume fraction.
Slope represents the density of the substance On a mass/volume graph, y = mass, x = volume, so y÷x = mass/volume, which = density Use the graph provided to answer the following 1a.which substance has a greater density

What type of slope did you look for to determine your answer?
Use the data found in the table on water, oil, and corn syrup to make a graph on the next page Use volume as your independent (manipulated) variable and mass as the dependent (responding) variable The volume of these substances can be any number (23 ml, 37.25 ml,.232 ml, etc.) so the data are continuous rather than discrete. When creating a mass vs volume graph in excel, it is important to start by understanding the data that will be used for the graph
This involves gathering the relevant mass and volume data and ensuring that it is clean and consistent. Lets say we need to k w the volume occupied by 50.0 g of this substance We would so (y−b) x= This energy is described by e = mc2, where c is the speed of light
In terms of density, m = ρv, where ρ is the volumetric mass density, v is the volume occupied by the mass
This energy can be released by the processes of nuclear fission (~ 0.1%), nuclear fusion (~ 1%), or the annihilation of. Learn how to calculate density using mass vs Volume graphs in just 5 minutes This video lesson walks you through the process with examples, followed by a quiz.
Graph the following mass and volume numbers on the graph below This graph is a line graph and will look like the graph on the other side of the homework Mass of liquid (g) 20 100 75 40 10 volume of liquid (cm3) 4 20 15 8 2 100 density of unknown substance line graph 90 80 mass (g) 70 60 50 40 30 20 10 0 2 4 6 8 10 12 14 16 18 20 22 volume (ml) 7. Show how to graph the mass and volume data for a material and then how to use the slope of the line on the graph to calculate the density

The density of a substance is its mass per unit volume.