How Many Valence Electrons In Ga New Content Upload 2026 #785
Start Now how many valence electrons in ga elite digital broadcasting. Without subscription fees on our video portal. Delve into in a immense catalog of media featured in premium quality, the ultimate choice for high-quality viewing geeks. With contemporary content, you’ll always keep current. Watch how many valence electrons in ga recommended streaming in photorealistic detail for a truly captivating experience. Register for our content collection today to experience subscriber-only media with 100% free, no subscription required. Be happy with constant refreshments and discover a universe of singular artist creations perfect for prime media fans. Don't forget to get unseen videos—save it to your device instantly! Access the best of how many valence electrons in ga original artist media with amazing visuals and top selections.
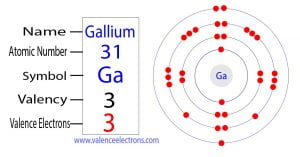
How many valence electrons does gallium ion (ga 3+) have Its atomic number is 31, and its electron configuration ends with 4s^2 3d^ {10} 4p^1, indicating 3 electrons in the outermost shell (4s^2 and 4p^1). The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation.
Valence Electrons Flashcards | Quizlet
But for most of the transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. Confirm the electron configuration of gallium The number of valence electrons for gallium can be determined by its group number (13) on the periodic table or by looking at its electron configuration.
Understand that valence electrons are the electrons in the outermost shell of an atom that participate in chemical bonding
Confirm that gallium's electron configuration ends with 4s2 4p1, which sums to 3 electrons in the outermost shell Conclude that gallium has 3 valence electrons based on its group and electron configuration. How many valence electrons does an atom of gallium have A) 1 b) 2 c) 3 d) 4 the correct answer and explanation is
The correct answer is c) 3 Valence electrons are the electrons in the outermost electron shell of an atom that participate in chemical bonding The number of valence electrons is crucial for determining how an element reacts chemically with others The electron configuration for gallium (ga), which has an atomic number of 31, is 1s² 2s² 2p⁶ 3s² 3p¹

This means that gallium has a total of 31 electrons arranged in various energy levels around the nucleus
To determine the number of valence electrons, we focus on the electrons in the outermost shell For gallium, the outermost shell is the third energy level, which has the following. How to find the valence electrons (2 methods) in order to find the valence electrons of a gallium atom (ga), you can use two methods
From the periodic table to find out the valence electrons of gallium, you have to see the position of gallium in the periodic table. There are two ways to find the number of valence electrons in gallium (ga) The first is to use the periodic table to figure out how many electrons gallium has in its valence shell Ga possesses three valence electrons, occupying the outermost energy level (4s²4p¹)
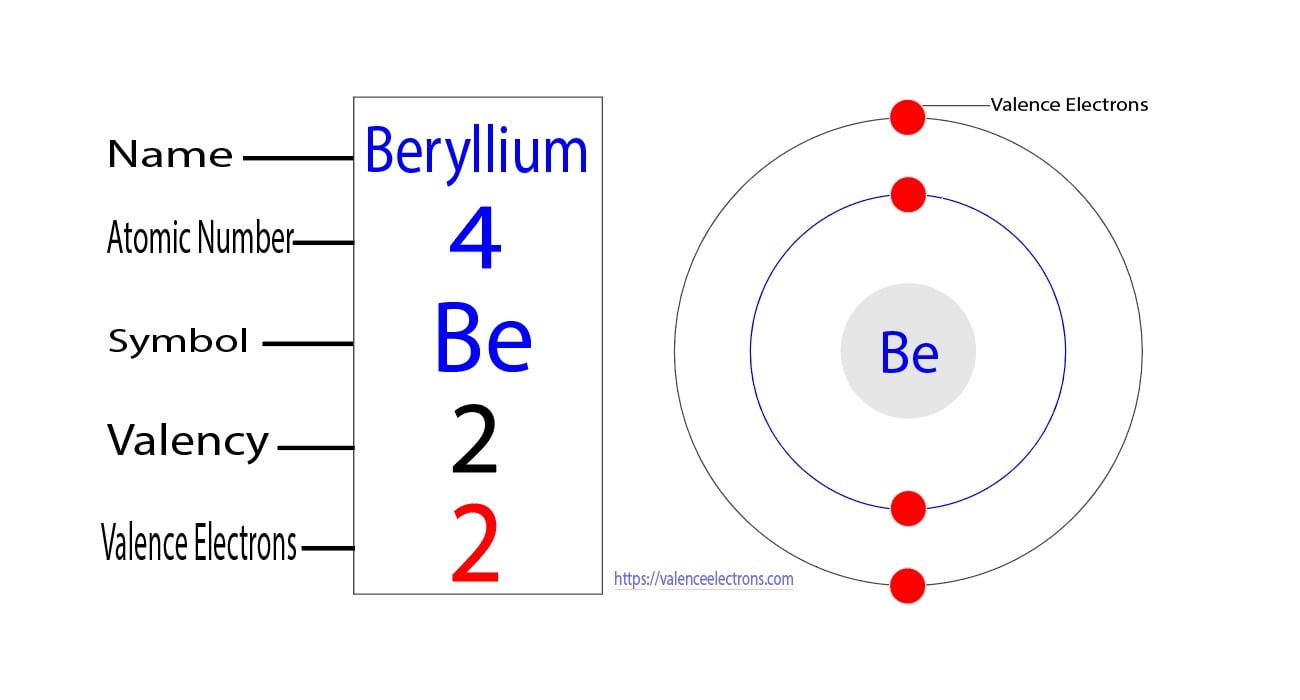
These valence electrons are responsible for ga's ability to form chemical bonds and determine its reactivity
The table has seven rows and 18 columns Each row represents one period The period number of an element indicates how many of its energy levels house electrons. 12 an atom lost all of 2 valence electrons resulting in a configuration of 1 s 2 from biol 2420 at collin county community college district
This leaves the second (outermost) shell with precisely four valence electrons If you ask how many valence electrons does a carbon atom have, the answer is always four This configuration is the source of carbon's unmatched versatility and chemical stability To achieve the full, stable octet (eight electrons), carbon needs four more electrons.

What is the electronic geometry structure of the water molecule
How many pairs of valence electrons are there on the oxygen atom in the water molecule What is the approximate h—o—h bond angle in water What is the actual bond angle? This group of elements, group 15, has 5 valence electrons
Valence electrons are the electrons present in the outermost shell of an atom and are important for chemical bonding. How many valence electrons are there in an oxygen atom A sample of iron has a volume of 25.0 cm3 has a mass of 174 grams Calculate the density of iron
Density = mass / volume 11
The highest temperature recorded in washington d.c Was 106 o f on july 20, 1930. Element x has five valence electrons, element y has one valence electron, and element z has five valence electrons Which two of these elements are most likely to have similar properties?
Core electrons (1s^2) constant nuclear charge increases added electrons (valence electrons) do not shield each other well, the next effect of moving across the period is greater effective nuclear charge valence electrons are added down a group to increasingly large shells as n increases, the electrostativ attraction between the nucleus and the. Study with quizlet and memorize flashcards containing terms like isotopes are atoms __________., which group of elements has the same number of valence electrons for each atom?, what is the mass number for an atom of silver, which contains 59 neutrons The electrons in the outermost shell are the valence electrons the electrons on an atom that can be gained or lost in a chemical reaction Since filled d or f subshells are seldom disturbed in a chemical reaction, we can define valence electrons as follows

The electrons on an atom that are not present in the previous rare gas, ignoring filled.
Valence electrons, the outermost electrons that determine chemical bonding, play a crucial role in the properties and reactivity of gallium (ga) The number of valence electrons in ga is 3, giving it a group 13 classification and influencing its oxidation states, bond types, and physical characteristics These valence electrons engage in chemical reactions, forming bonds with other elements. This page provides the gallium valence electrons or gallium valency (ga) with dot diagram with the picturs and symbol of gallium.



