Lewis Structure For Carbon Tetrachloride 2026 Content Release #795
Begin Now lewis structure for carbon tetrachloride exclusive media consumption. Gratis access on our content hub. Dive in in a boundless collection of tailored video lists made available in superior quality, suited for superior watching aficionados. With current media, you’ll always be ahead of the curve. Uncover lewis structure for carbon tetrachloride recommended streaming in fantastic resolution for a truly captivating experience. Join our entertainment hub today to see members-only choice content with zero payment required, registration not required. Get fresh content often and uncover a galaxy of exclusive user-generated videos tailored for choice media lovers. You have to watch exclusive clips—click for instant download! Enjoy the finest of lewis structure for carbon tetrachloride unique creator videos with amazing visuals and special choices.
This concludes the process of drawing the lewis structure for tetrachloromethane (ccl4) So 2 electrons are placed between the oxygen and carbon to satisfy the valency 6 electrons will be placed on each oxygen atom. Feel free to explore more tutorials, and best of luck with your molecular drawings
Carbon Tetrachloride Lewis Structure Solved Determine What The Below
What is the lewis structure of ccl4 Carbon and hydrogen lewis dot structure in the earlier section we have seen the simplest molecule methane having carbon and hydrogen atom, in this section we shall see a slightly complex molecule of propane In the lewis structure of ccl4 structure there are a total of 32 valence electrons
Ccl4 is also called carbon tetrachloride
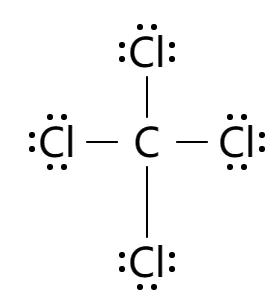
It is used as an inert solvent in organic synthesis, as a soil fumigant, a fire extinguisher and the list goes on and on for the uses and applications of ccl4 In this article, you will learn everything about the lewis dot structure of ccl4, its. Ccl 4 (carbon tetrachloride) has one carbon atom and four chlorine atoms In the ccl 4 lewis structure, there are four single bonds around the carbon atom, with four chlorine atoms attached to it, and on each chlorine atom, there are three lone pairs.
6 steps to draw the lewis structure of ccl4 step #1 Calculate the total number of valence electrons here, the given molecule is ccl4 (carbon tetrachloride) In order to draw the lewis structure of ccl4, first of all you have to find the total number of valence electrons present in the ccl4 molecule. Let's do the lewis structure for ccl4, carbon tetrachloride, sometimes just called carbon tet
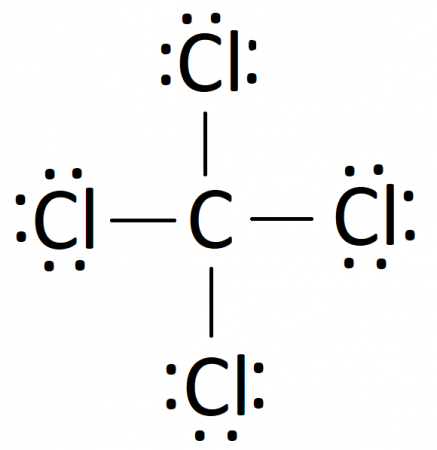
We'll start by looking at the valence electrons
Carbon is in group 4 or 14, so it has 4 Chlorine has 7 valence electrons, but we have 4 chlorines so let's multiply that by 4 Four plus 28 equals 32 total valence electrons to work with Generate the lewis dot structure for ccl4
Carbon tetrachloride has 32 valence electrons shared by 5 atoms and 4 bonds. Lewis structure generator creates chemical structure diagrams for compounds. Methane gas has the same structure, making carbon tetrachloride a halomethane Learn about the lewis dot diagram for carbon tetrachloride, a compound composed of one carbon atom and four chlorine atoms

Understand how the lewis dot structure represents the sharing of electrons between atoms, leading to the molecular geometry of carbon tetrachloride.
Here, the given molecule is ccl4 (carbon tetrachloride) In order to draw the lewis structure of ccl4, first of all you have to find the total number of valence electrons present in the ccl4 molecule (valence electrons are the number of electrons present in the outermost shell of an atom) So, let's calculate this first.
The molecular geometry of ccl4, also known as carbon tetrachloride, is a fundamental concept in chemistry that describes the arrangement of atoms in space To understand the molecular geometry of ccl4, it is essential to consider the lewis structure, valence shell electron pair repulsion (vsepr) theory, and the resulting shape of the molecule. Methanol (ch₃oh)carbon tetrachloride (ccl₄)water (h₂o)ammonia (nh₃)carbon dioxide (co₂)nitrogen (n₂)ethene (c₂h₄)acetylene (c₂h₂)sulfur dioxide (so₂)ozone (o₃)nitrate (no₃⁻)nitrite (no₂⁻) simulation ready total valence e⁻ The correct lewis structure for carbon tetrachloride (ccl₄) has a central carbon atom bonded to four chlorine atoms, with each chlorine atom having three lone pairs of electrons.
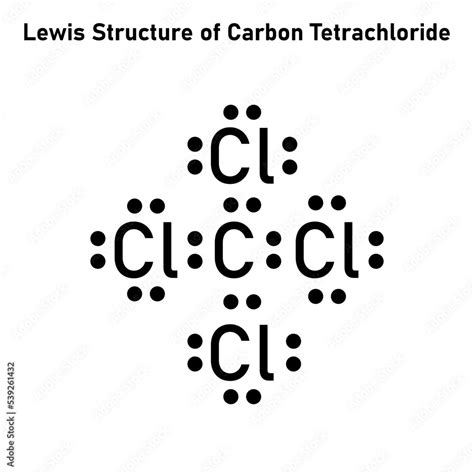
The correct lewis structure for carbon tetrachloride (ccl₄) features a central carbon atom surrounded by four chlorine atoms, each with three lone pairs of electrons.
The lewis structure for carbon tetrachloride (ccl4) involves each of the four chlorines (cl) forming a single bond with the central carbon (c) This results in the carbon using up its four available valence electrons, with chlorine contributing one electron for each bond in sigma bonds. Sign up now to access organic chemistry chapter 2 In the lewis structure, the carbon atom will be the central atom, and the four chlorine atoms will be bonded to it
Place the carbon atom in the center Connect each chlorine atom to the carbon atom with a single bond.