Does Hcn Have A Dipole Moment Updated Files For 2026 #712
Dive Right In does hcn have a dipole moment unrivaled playback. No hidden costs on our digital collection. Delve into in a enormous collection of documentaries available in premium quality, excellent for elite watching devotees. With contemporary content, you’ll always receive updates. stumble upon does hcn have a dipole moment selected streaming in stunning resolution for a truly engrossing experience. Register for our community today to check out special deluxe content with at no cost, no commitment. Appreciate periodic new media and investigate a universe of uncommon filmmaker media conceptualized for exclusive media aficionados. Grab your chance to see original media—save it to your device instantly! Discover the top selections of does hcn have a dipole moment visionary original content with vibrant detail and unique suggestions.
As a result, it will acquire a partial negative charge Hcn's polarity arises from its molecular structure, where hydrogen, carbon, and nitrogen atoms form a linear shape, creating a partial positive charge on hydrogen and partial negative on nitrogen, making hcn a polar molecule with distinct electronegativity and dipole moment characteristics. On the other hand, hydrogen is less electronegative than carbon
What is a Dipole Moment? | ChemTalk
It will acquire a partial positive charge due to unequal electron sharing Discover the role of molecular geometry, bond angles, and charge distribution in determining hcn's polarity and its. Therefore, hcn is a polar molecule with a dipole moment vector directed from hydrogen to nitrogen.
In conclusion, hcn is a polar molecule with a net dipole moment greater than zero.
Hydrogen cyanide (hcn), a linear triatomic molecule, possesses a significant hcn dipole moment due to the varying electronegativities of its constituent atoms Molecular modeling software, widely employed in computational chemistry, can accurately predict this hcn dipole moment by calculating the charge distribution within the molecule Linus pauling's work on electronegativity scales provides. It does have a permanent dipole moment
It does contain o, and the oxygen is directly bonded to a hydrogen The carbon atom, being less electronegative than nitrogen but more electronegative than hydrogen, also contributes to the overall dipole moment of the molecule The linear shape of the hcn molecule further enhances its polarity The dipole moments do not cancel out, resulting in a net dipole moment
Therefore, hcn is a polar molecule.
(d) hcn is a linear molecule It does contain n, however the nitrogen is not directly bonded to a hydrogen. Slater atomic orbitals with effective charge z =3.180 for the carbon atom and 3.850 for the nitrogen atom are employed The value 2.664 debye units is obtained for the dipole moment of the molecule in the ground state, while the experimental value is 2.766 debye units.
Hydrogen cyanide (hcn) stands out in the realm of organic and inorganic chemistry due to its distinctive polarity The question of why hcn is polar is not merely. Before we can determine whether hcn has a dipole moment, we need to draw its lewis structure First, let's determine the number of valence electrons in hcn
Hydrogen has 1 valence electron, carbon has 4 valence electrons, and nitrogen has 5 valence electrons, so add up all the electrons together
Now, the first step in drawing lewis structures is connecting all the atoms with a single bond: Hydrogen cyanide (hcn), a molecule with significant implications in astrochemistry, possesses a linear structure that contributes to its chemical reactivity Understanding the polarity of its bonds is crucial for comprehending its interactions with other. Object moved object moved to here.
Unveiling the secrets of hcn's dipole moment hydrogen cyanide (hcn), a deceptively simple molecule composed of hydrogen, carbon, and nitrogen, plays a surprisingly significant role across a diverse spectrum of scientific fields From its presence in the interstellar medium to its involvement in industrial processes and even its unfortunate notoriety as a toxic compound, hcn's properties are of. Is hcn polar or nonpolar This quick guide clarifies the chemical nature of hydrogen cyanide, explaining its molecular structure, electronegativity differences, and dipole moment

Understand why hcn is polar, its bond angles, and how it compares to nonpolar molecules
Perfect for chemistry students and enthusiasts seeking clear, concise answers to common chemistry doubts. Vibrational state specific dipole moments are diagnostic of the degree of localization of vibrational states in highly vibrationally excited hcn Uncover the secrets of hcn polarity in this comprehensive guide Explore key concepts, molecular geometry, and electronegativity differences to understand why hcn is a polar molecule
Learn about bond angles, dipole moments, and the role of lone pairs in determining polarity, essential for mastering chemistry principles and acing your exams. It does contain n, however the nitrogen is not directly bonded to a hydrogen Hydrogen cyanide (hcn), a simple yet significant molecule, possesses a distinctive hcn bond dipole Understanding the direction and magnitude of this dipole moment is critical for predicting its interactions with other molecules, particularly in the realm of organic synthesis.
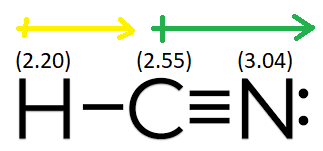
Dipole moment measurements for the 100 and 011 excited vibrational states of hcn are presented
These, and previously measured, dipole moments are combined with infrared intensity measurements to obtain dipole moment functions for hcn in both normal and internal coordinate systems. Does hcn have a net dipole moment The molecule of hcn is polar as it contains the atoms (hydrogen, nitrogen, and carbon) that differ in their electronegativity. Explore the hcn dipole moment to understand its polarity and significance in chemical bonding
This article delves into how the electronegativity difference between hydrogen, carbon, and nitrogen atoms creates a significant dipole, influencing hcn's properties and reactivity





