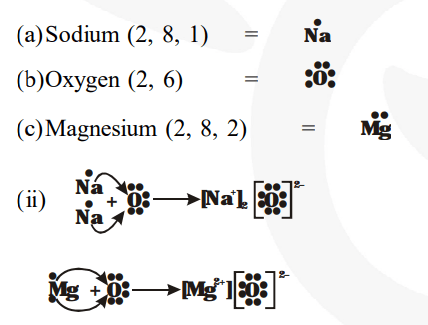Electron Dot Structure For Mg Original Creator Submissions #921
Enter Now electron dot structure for mg signature playback. Pay-free subscription on our cinema hub. Become one with the story in a massive assortment of themed playlists featured in superb video, the best choice for superior watching viewers. With current media, you’ll always remain up-to-date. Witness electron dot structure for mg arranged streaming in breathtaking quality for a truly engrossing experience. Enter our digital stage today to enjoy VIP high-quality content with 100% free, registration not required. Look forward to constant updates and browse a massive selection of singular artist creations conceptualized for superior media supporters. Act now to see one-of-a-kind films—rapidly download now! Explore the pinnacle of electron dot structure for mg unique creator videos with vivid imagery and members-only picks.
Generate the lewis dot structure for mg Find the lewis dot structure for magnesium in this video. Enter a chemical element or formula to calculate and draw its lewis dot structure
Electron Dot Structure For Magnesium
Be sure to use the proper capitalization for all element symbols For determining its lewis structure, we first look at its valence electrons For the lewis structure of individual elements, use our valence electron calculator
The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge.
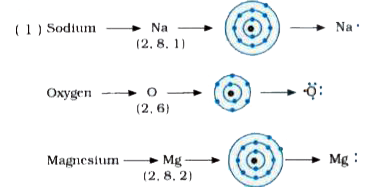
I show you where magnesium is on the periodic table and how to determine how many valence electrons magnesium has. The electron dot diagram for magnesium (mg) represents the arrangement of its valence electrons, which are the electrons in the outermost energy level The diagram is a way to visualize the bonding and valence electrons of an atom, and it helps to understand the chemical properties of elements. A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. There are two valence electrons in mg atom Hence, the lewis dot symbol for mg is
There is only one valence electron in an atom of sodium
Hence, the lewis dot structure is There are 3 valence electrons in boron atom There are six valence electrons in an atom of oxygen Learn how to draw the lewis dot diagram for magnesium and understand its electronic structure.
The lewis structure for mg (magnesium) shows two valence electrons surrounding the magnesium atom This representation highlights magnesium's position as an alkaline earth metal, which readily loses these two electrons to form a mg²+ ion. Web, a lewis electron dot diagram (or electron dot diagram, or a lewis diagram, or a lewis structure) is a representation of the valence electrons of an atom that uses Its electron dot diagram is as follows test yourself what is the lewis electron dot diagram for each element

Magnesium oxide consists of magnesium and oxygen and has the formula mgo, which consist of ionic bonds
The electron dot structure of chlorine is given below When 2 chlorine atoms and magnesium atoms combine by transfer of electrons then magnesium chloride is formed by transfer of electrons Chlorine atom needs only one electron to complete its octet and magnesium cal removes two electrons to complete its octet. It is a group two and period three element