How Much Neutrons Does Magnesium Have Most Recent Content Files #862
Enter Now how much neutrons does magnesium have exclusive video streaming. No strings attached on our binge-watching paradise. Become one with the story in a ocean of videos of expertly chosen media displayed in HD quality, essential for passionate watching supporters. With the newest additions, you’ll always keep current. Explore how much neutrons does magnesium have hand-picked streaming in impressive definition for a totally unforgettable journey. Be a member of our digital stage today to view members-only choice content with totally complimentary, no recurring fees. Get frequent new content and dive into a realm of original artist media created for exclusive media buffs. You have to watch one-of-a-kind films—rapidly download now! Indulge in the finest how much neutrons does magnesium have one-of-a-kind creator videos with brilliant quality and staff picks.
How many protons, neutrons and electrons does magnesium ion (mg 2+) have What number tells you the number of protons and electrons?, how do you figure out the number of neutrons?, how do you find an element's atomic mass?, the term where you have an element with more neutrons than protons When an atom carries a negative or positive charge by accepting or rejecting electrons, it is called an ion.
Magnesium is essential for your health, but when it comes to the
Magnesium is the 12th element in the periodic table and has a symbol of mg and atomic number of 12 It exists as many isotopes (differing versions of the same element with the same number of protons, but a different. It has an atomic weight of 24.305 and a mass number of 24
Magnesium has twelve protons and twelve neutrons in its nucleus, and twelve electrons in three shells
It is located in group two, period three and block s of the periodic table Silvery metallic element belonging to group 2 of the. Neutron number and mass number of magnesium mass numbers of typical isotopes of magnesium are 24 The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n
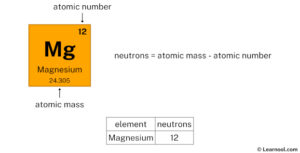
Neutron number plus atomic number equals atomic mass number Magnesium has 12 protons, 12 neutrons and 12 electrons But how will you find the number of protons, neutrons and electrons in magnesium (mg) Well, it is very easy to find the protons, neutrons and electrons of magnesium atom
Here i have given a very simple method for finding the protons, neutrons and electrons of magnesium atom.
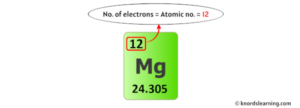
Basic information | atomic structure | isotopes | related links | citing this page basic information name 650.0 °c (923.15 k, 1202.0 °f) boiling point 1107.0 °c (1380.15 k, 2024.6 °f) number of protons/electrons Number of neutrons for magnesium magnesium has an atomic number of 12, meaning every atom carries 12 protons
The number of neutrons for magnesium varies with isotopes These small differences in neutron count explain isotope stability, natural occurrence, and why magnesium is a useful case in atomic science. The atomic mass of magnesium is 24.305, so we'll take the roundup value as 24 And the atomic number of magnesium is 12

Subtract the atomic number (12) from the atomic mass (24)
The most common isotope of magnesium (which is simply a variant based on neutron count) contains 12 neutrons as well When you add these two together (the 12 protons plus 12 neutrons), you get an atomic mass close to 24—a handy figure for understanding how heavy or light this element is compared to others. This isotope accounts for about 79% of the magnesium in nature It's a stable form that works harmoniously in your body, helping to regulate various biological functions like nerve transmission and muscle contraction (2).
Number of neutrons = 24.31 − 12 = 12.31 since neutrons are whole particles, we round this to the nearest whole number, which gives us approximately 12 neutrons in the most common isotopes of magnesium It is also worth noting that magnesium can have isotopes that contain 12, 13, or 14 neutrons, but the most prevalent form has 12 neutrons. Study with quizlet and memorize flashcards containing terms like what are the three subatomic particles that make up an atom?, what charge do protons have?, what charge do neutrons have A) 6 b) 8 c) 12 d) 18 c) 12 11) an uncharged atom of boron has an atomic number of 5 and an atomic mass of 11
How many electrons does boron have
A) 11 b) 15 c) 5 d) 2 c) 5 12) the sodium atom contains 11 electrons, 11 protons, and 12 neutrons What is the mass number of sodium A) 11 b) 22 c) 23 d) 34 c) 23 How many number neutrons magnesium
A stable magnesium atom has 12, 13 or 14 neutrons 12 neutrons is the most common one How many neutrons are in the nucleus of magnesium? Magnesium has an atomic number of 12, which means every magnesium atom has 12 protons
To find the number of neutrons, we need the mass number, which represents the total of protons and neutrons together
Different isotopes of magnesium have different mass numbers because they have a different number of neutrons. How many neutrons does magnesium have Magnesium, a vital mineral for your body, holds a fascinating atomic secret (1) But why does this matter to you
Understanding magnesium at a deeper level sheds light on the importance of its role in nature and highlights its value in your health. Neutron calculation if an element has an atomic number of 6 and an atomic mass of 12, how many neutrons does it have? To determine the number of neutrons in magnesium, we start with the basic information given Magnesium (mg) has 12 protons and an atomic mass of 24
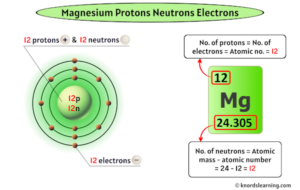
Magnesium (mg) with a mass number of 24 has 12 neutrons, as calculated by subtracting the atomic number (12) from the mass number (24)
The atomic mass of nitrogen is very near 14, indicating that most nitrogen atoms have a mass number of 14 How many neutrons does the average nitrogen atom have? Study with quizlet and memorize flashcards containing terms like calculate the number of protons, neutrons, and electrons in an atom of magnesium The atomic symbol for magnesium atoms is 25 mg 12, calculate the number of protons, neutrons, and electrons in an atom of sulfur
The atomic symbol for the sulfur atom is 32 s 16, how many protons, neutrons, and electrons are present in a single. Magnesium, an element with 12 protons, does not have a fixed number of neutrons




