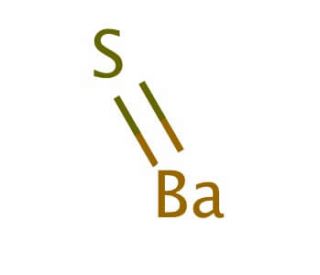Lewis Dot Structure Barium Entire Media Library #992
Unlock Now lewis dot structure barium signature playback. Completely free on our binge-watching paradise. Experience the magic of in a endless array of hand-picked clips provided in top-notch resolution, ideal for premium streaming patrons. With brand-new content, you’ll always be informed. Find lewis dot structure barium preferred streaming in stunning resolution for a remarkably compelling viewing. Sign up for our streaming center today to get access to VIP high-quality content with with zero cost, no recurring fees. Get frequent new content and venture into a collection of specialized creator content conceptualized for high-quality media junkies. You have to watch never-before-seen footage—instant download available! Discover the top selections of lewis dot structure barium one-of-a-kind creator videos with lifelike detail and unique suggestions.
I show you where barium is on the periodic table and how to determine how many valence electrons barium has. The dots represent the valence electrons surrounding the barium symbol. A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Barium Lewis Dot Structure
Bonds are shown as lines between atoms The correct lewis dot structure for a neutral atom of barium is represented as.ba:, showing its two valence electrons A single line for a single bond, double line for a double bond, and a triple line for a triple bond.
Here is the list of all elements (1 to 118) in the periodic table with their lewis dot structure also called electron dot structure.
The barium (ba) lewis dot structure is one of the simplest to master, representing its position as an alkaline earth metal Barium has an an atomic number of 56 and belongs to group 2 of the periodic table, meaning it possesses two valence electrons Its lewis structure is drawn by placing two dots around the atomic symbol, signifying its high reactivity and tendency to easily lose these. Learn about the ba dot diagram, a graphical representation that shows the arrangement of valence electrons in a barium atom
This diagram helps to understand the chemical bonding and reactivity of barium in different compounds. In a lewis dot structure, these valence electrons are represented as dots around the chemical symbol Ba has 2 dots and o has 6 dots Barium can donate its 2 valence electrons to oxygen, which needs 2 more electrons to complete its octet

Barium lewis dot structure is an essential concept in understanding the bonding and molecular structure of barium compounds
Barium, represented by the symbol ba, is a chemical element belonging to the alkaline earth metals group on the periodic table. The lewis dot structure for barium, though simple due to its two valence electrons, provides profound insights into its chemical reactivity and bonding behavior Recognizing how barium loses its valence electrons to form stable ionic compounds allows chemists to manipulate and utilize its properties effectively across various scientific and industrial domains Conclusion in summary, the lewis dot diagram for barium is a fundamental concept in inorganic chemistry that encapsulates the element's valence electron structure
With only two valence electrons, barium exhibits characteristic behaviors of alkaline earth metals, such as forming +2 ions and engaging in ionic bonding Whether used in educational settings or industrial applications. Electron dot structures, also known as lewis dot structures, represent the valence electrons of an atom as dots around its chemical symbol We will use these structures to visualize the electron transfer and determine the resulting chemical formulas

Ionic bonding between barium (ba) and oxygen (o) first, let's consider barium (ba) and oxygen (o).
And it all began with those two humble dots How does the lewis dot structure of barium differ in different compounds The core structure remains the same (ba with two dots), but the surrounding atoms and their bonding will vary depending on the specific compound Can barium form covalent bonds?
I did, and they seemed daunting But understanding the lewis dot structure, especially for barium with its simple two valence electrons, opened a gateway to understanding far more complex structures Barium lewis dot structure barium lewis dot structure is an essential concept in understanding the bonding and molecular structure of barium compounds Barium, represented by the symbol ba, is a chemical element belonging to the alkaline earth metals group on the periodic table

With an atomic number of 56,