Aluminum Hydrochloric Acid Balanced Equation Original Video Content #938
Unlock Now aluminum hydrochloric acid balanced equation top-tier broadcast. Completely free on our viewing hub. Submerge yourself in a vast collection of tailored video lists demonstrated in top-notch resolution, great for high-quality streaming enthusiasts. With hot new media, you’ll always never miss a thing. Reveal aluminum hydrochloric acid balanced equation curated streaming in amazing clarity for a genuinely engaging time. Participate in our network today to get access to unique top-tier videos with with zero cost, no sign-up needed. Benefit from continuous additions and experience a plethora of uncommon filmmaker media conceptualized for select media buffs. This is your chance to watch singular films—download immediately! Discover the top selections of aluminum hydrochloric acid balanced equation specialized creator content with lifelike detail and top selections.
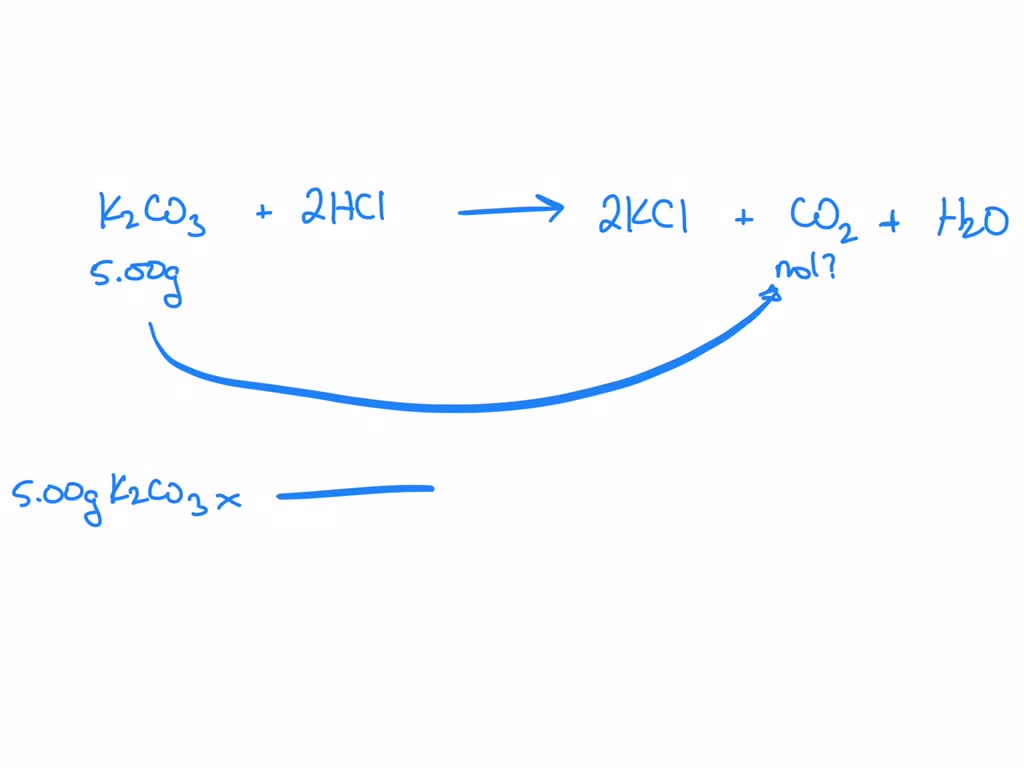
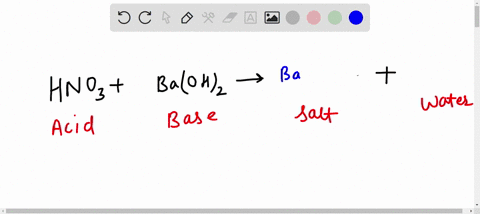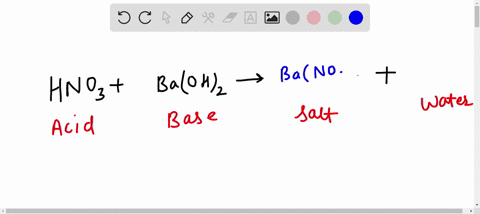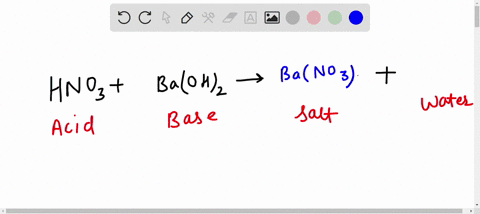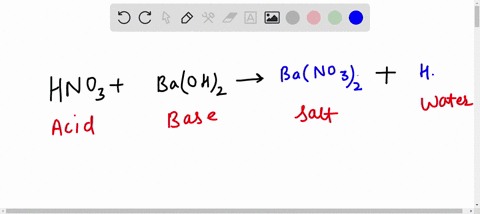
Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced What is the balanced equation for aluminum and hydrochloric acid 2 al + 6 hcl = 2 alcl3 + 3 h2
SOLVED: In the balanced equation for acid reflux (assume hydrochloric
Instructions on balancing chemical equations To balance the reaction between solid aluminum and hydrochloric acid, we need to ensure that the number of atoms of each element is the same on both sides of the equation. Enter an equation of a chemical reaction and click 'balance'
The answer will appear below always use the upper case for the first character in the element name and the lower case for the second character
Fe, au, co, br, c, o, n, f Write the balanced chemical equation for the reaction of aluminium (al) with hydrochloric acid (hcl) The products are aluminium chloride (alcl₃) and hydrogen gas (h₂). Aluminum (al) reacts with hydrochloric acid (hcl) to form aluminum chloride (alcl3) and hydrogen gas (h2)
The unbalanced chemical equation is Al+hcl→alcl3+h2 we will balance the equation by adjusting coefficients so that the number of atoms of each element is the same on both sides Identify the reactants and products Aluminum (al) reacts with hydrochloric acid (hcl) to produce aluminum chloride (alcl 3) and hydrogen gas (h 2)
Al + hcl → alcl 3 + h 2 balance the equation
Balance the number of atoms of each element on both sides of the equation by adjusting the stoichiometric coefficients. The balanced equation for the reaction between solid aluminum and hydrochloric acid is 2 al (s) + 6 hcl (aq) → 2 alcl₃ (aq) + 3 h₂ (g) This equation represents the production of aluminum chloride and hydrogen gas from the reactants. Al + hcl = alcl3 + h2
Solved and balanced chemical equation Online calculator for equalizing chemical reactions. For balancing any chemical equation, we have to maintain equal molecularity of each atom present on the reactant side as well as on the product side of the given chemical reaction Complete step by step solution
Given that, aluminum and hydrochloric acid reacts together to form aluminum chloride and hydrogen gas and chemical reaction for this is shown as follow
Al + hcl → alc l 3 + h. Based upon your experimental results, write a correctly balanced equation for the reaction of aluminum with hydrochloric acid The reaction between aluminum and hydrochloric acid is a fascinating and important chemical reaction with a variety of applications Understanding the balanced chemical equation, the underlying chemistry, and the safety precautions associated with this reaction is essential for anyone working with these chemicals.
With an aid of well balanced equation write a flow diagram of making sulphuric acid (100 marks? Write a balanced equation copper (ii) oxide solid with dilute sulphuric (vi)acid?? From the balanced equation, we can see that 1 molecule of aluminum hydroxide (al (oh)₃) reacts with 3 molecules of hydrochloric acid (hcl) for complete neutralization. A sample of is ignited 2c₁₀h₁₈ 18h2o 49
Aluminum metal is placed into a test tube of hydrochloric acid 2a1 6hc1 2aici, 50
Tetraphosphorus decaoxide is mixed with water p4o10 ut2 4h3po4 4 directions Estimate enthalpy changes for each of the following reactions applying bond energy calculations. Aluminum reacts with hydrochloric acid to produce aluminum chloride and hydrogen Write a balanced equation for the reaction
Calculate the number of moles of hydrochloric acid required to react with 0.87 mole of aluminum. An iron nail is dipped in copper two sulphate solution a Write two things that you would observe b Construct a balanced equation c
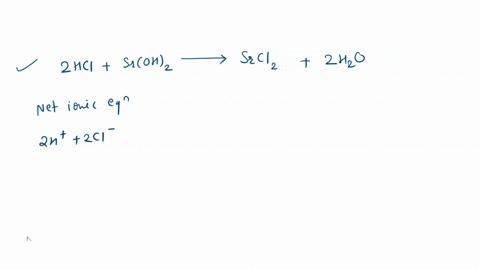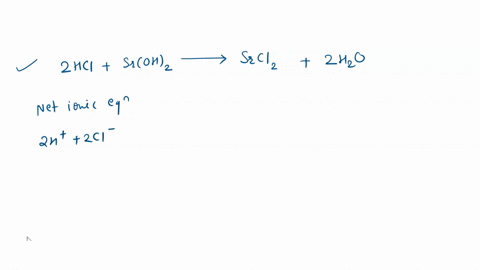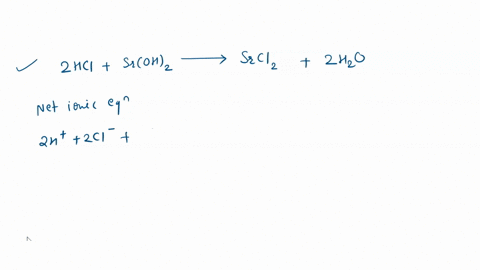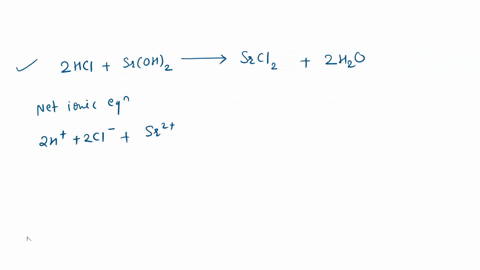
Write the balanced equation for the reaction of aluminum metal with hydrochloric acid
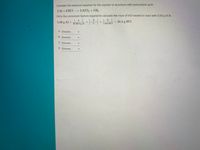
The coefficients on the aluminum metal and the hydrochloric acid are 2 al (s) and 6 hcl (aq) 6 al (s) and 2 hcl (aq) 1 al (s) and 2 hcl (aq) 3 al (s) and 6 hcl (aq) o 3 al (s) and 2 hcl (aq) • previous next 10:59 backs & 96 * 00 9 5 6 7 n This video discusses how to write the balanced molecular equation between aluminum and hydrochloric acid It also shows you how to write the net ionic equation between al and hcl.
Click here:point_up_2:to get an answer to your question :writing_hand:how can i balance this chemical equations aluminum and hydrochloric acid react to form aluminum