How Many Electrons Does Magnesium Have In Its Outer Shell Photo & File Content Updates #679
Begin Your Journey how many electrons does magnesium have in its outer shell select viewing. No recurring charges on our content hub. Become one with the story in a boundless collection of series demonstrated in superb video, great for elite watching aficionados. With recent uploads, you’ll always keep current. stumble upon how many electrons does magnesium have in its outer shell organized streaming in fantastic resolution for a deeply engaging spectacle. Link up with our platform today to enjoy VIP high-quality content with no charges involved, no need to subscribe. Get frequent new content and uncover a galaxy of groundbreaking original content developed for elite media enthusiasts. Grab your chance to see specialist clips—download quickly! Enjoy the finest of how many electrons does magnesium have in its outer shell exclusive user-generated videos with exquisite resolution and top selections.
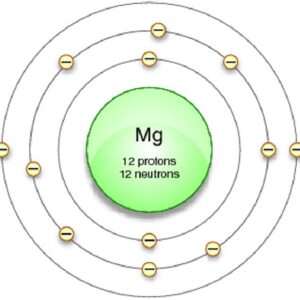
Thus, 1st shell can hold 2 electrons Looking at the picture, you can see there are two electrons in shell one, eight in shell two, and two more in shell three. 2nd shell can hold 8 electrons
How many valence electrons does magnesium have - Mixsaver
3rd shell can hold 18 electrons That means there are 12 electrons in a magnesium atom 4th shell can hold 32 electrons
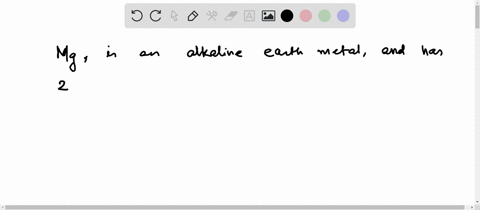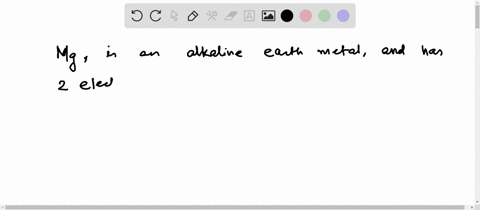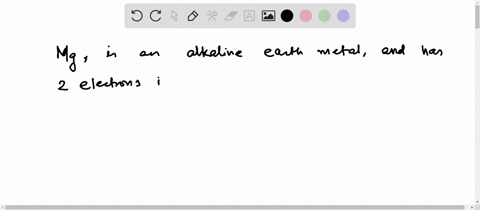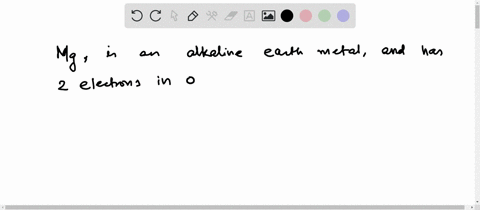
Now the atomic number of magnesium (mg) is 12
Hence the magnesium element has electrons arrangement 2, 8, 2 This electron arrangement indicates that the outermost orbit of magnesium element (mg) has 2 electrons. Magnesium has 2 electrons in its outer shell This information comes from its electron configuration, which indicates that its outermost shell, the third one, contains 2 electrons
Understanding electron configuration is essential in chemistry for predicting element behavior. The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation Magnesium donates the electron of the last shell to form bonds and turns into a magnesium ion (mg +2). Magnesium has a total of 12 electrons

The electron configuration would be 1s2 2s2 2p6 3s2
Therefore, 2 electrons in it's outer shell. Magnesium number of valence electrons there are two valence electrons in the outer shell of the magnesium Many other valence electrons of the element have been available here Hydrogen valency helium valency lithium valency beryllium.
This electron configuration of magnesium shows that the outer shell of magnesium has just 2 electrons (3s2), hence, the number of valence electrons in the magnesium atom is 2. How to write the electron configuration for magnesium (mg) in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons) When we write the configuration we'll put all 12 electrons in orbitals around the nucleus of the magnesium atom. Does magnesium have a full outer shell of electrons
Magnesium has two electrons in its outer shell as it is in group 2 of the periodic table, which it loses, so its outer shell is full (2,8) and it has a charge of +2, as it still has 12 protons (positive charges) and now has only 10 electrons (negative charges).
Does magnesium have 3 electron shells In an atom, the electrons spin around the center, also called the nucleus